Health
CDC Issues New Guidelines, Launches Probe After 1000s Negatively-Affected Following COVID-19 Vaccination
… the agency stressed that anyone who has ever had a severe allergic reaction to any ingredient in a COVID-19 vaccine should not get that vaccine.

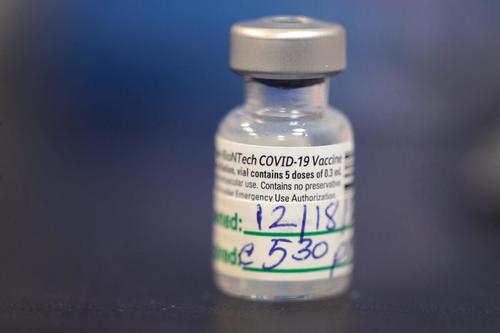
Thousands of people have been unable to work or perform daily activities, or required care from a healthcare professional, after getting the new COVID-19 vaccine, according to new data from the Centers for Disease Control and Prevention (CDC).
As of Dec. 18, 3,150 people reported what the agency terms “Health Impact Events” after getting vaccinated.
The definition of the term is: “unable to perform normal daily activities, unable to work, required care from doctor or health care professional.”
As The Epoch Times’ Zachary Stieber reports, the people reporting the negative effects reported them through V-safe, a smartphone application. The tool uses text messages and web surveys to provide personalized health check-ins and allows users to quickly tell the CDC if they are experiencing side effects.
The CDC and Pfizer, which produces the vaccine with BioNTech, didn’t respond to request for comments.
The information was presented by Dr. Thomas Clark, a CDC epidemiologist, to the Advisory Committee on Immunization Practices, an independent panel that provides recommendations to the agency, on Saturday.
The CDC said that 272,001 doses of the vaccine were administered as of Dec. 19. That means most people who were vaccinated did not experience negative effects.
The CDC has identified six case reports of anaphylaxis, or severe allergic reaction, that occurred following vaccination with the new vaccine, Clark reported. Other case reports were reviewed and determined not to be of anaphylaxis.
In an update on Friday, the agency stressed that anyone who has ever had a severe allergic reaction to any ingredient in a COVID-19 vaccine should not get that vaccine. People with severe allergic reactions to other vaccines should consult their doctor about getting the new vaccine while those with a history of anaphylaxis not related to vaccines “may still get vaccinated.”
“CDC recommends that people with a history of severe allergic reactions not related to vaccines or injectable medications – such as allergies to food, pet, venom, environmental, or latex – may still get vaccinated,” the CDC said.
“People with a history of allergies to oral medications or a family history of severe allergic reactions, or who might have a milder allergy to vaccines (no anaphylaxis) – may also still get vaccinated.”
Anyone who experiences anaphylaxis after getting the first vaccine should not get the second shot, the CDC said. COVID-19 vaccines are meant to be given across two doses, spaced about three weeks apart.
At least five healthcare workers in Alaska experienced adverse reactions after getting the Pfizer vaccine, the Anchorage Daily News reported. One of two experiencing adverse reactions at the Bartlett Regional Hospital required treatment at the hospital for at least two nights.
An Illinois hospital halted vaccinations after four workers suffered adverse reactions.
Dr. Peter Marks, the director of Food and Drug Administration’s Center for Biologics Evaluation and Research, told reporters in a call on Thursday night that the agency is working with the CDC, and colleagues in the United Kingdom, on probing the allergic reactions.
“We’ll be looking at all of the data we can from each of these reactions to sort out exactly what happened. And we’ll also be looking to try to understand which components of the vaccine might be helping to produce them,” he said.
Noting that he was speculating, Marks said it’s known that polyethylene glycol – a component present in both the Pfizer vaccine and one from Moderna that regulators approved earlier in the day – can be associated, uncommonly, with allergic reactions.
“So that could be a culprit here. And that’s why we’ll be watching very closely,” he said. “But we just don’t know at this point.”
Both vaccines have “systemic side effects,” which are “generally mild,” Marks said.
They go away after a day. According to the FDA website, the most commonly reported side effects include tiredness, headache, muscle pain, and chills. The agency said they go away after several days.
One volunteer in Pfizer’s late-stage clinical trial experienced an allergic reaction. Two people in Moderna’s phase 3 clinical trial experienced anaphylactic reactions, the company said during a meeting on Thursday. But the data showed the benefits outweigh the risk, FDA officials said, as they granted emergency use authorization to the vaccines about seven days apart.
People who get a COVID-19 vaccine should be monitored for at least 15 minutes after getting vaccinated, according to the CDC.
If someone experiences a severe allergic reaction against getting a COVID-19 vaccine, vaccination providers are supposed to provide rapid care and call for emergency medical services. The person should continue to be monitored in a medical facility for at least several hours.
Health
FDA Committee Members Reviewing Pfizer Vaccine For Children Have Worked For Pfizer, Have Big Pfizer Connections
“The industry defends the attempts to influence committee members as simply efforts to best present their case”
The FDA’s Vaccines and Related Biological Products Advisory Committee is holding a virtual meeting Tuesday October 26 to discuss authorizing a Pfizer-BioNTech Coronavirus vaccine for children between the ages of 5 to 11 years old.
This committee has a lot of sway with the FDA and their findings will be relevant, considering the Biden administration is getting ready to ship vaccines to elementary schools and California has already mandated the vaccine for schoolchildren pending federal authorization.
But the meeting roster shows that numerous members of the committee and temporary voting members have worked for Pfizer or have major connections to Pfizer.
Members include a former vice president of Pfizer Vaccines, a recent Pfizer consultant, a recent Pfizer research grant recipient, a man who mentored a current top Pfizer vaccine executive, a man who runs a center that gives out Pfizer vaccines, the chair of a Pfizer data group, a guy who was proudly photographed taking a Pfizer vaccine, and numerous people who are already on the record supporting Coronavirus vaccines for children. Meanwhile, recent FDA Commissioner Scott Gottlieb is on Pfizer’s board of directors.
HERE’S THE MEETING ROSTER: Vaccines and Related Biological Products Advisory Committee October 26, 2021 Meeting Draft Roster.
Acting Chair Arnold S. Monto was a paid Pfizer consultant as recently as 2018.
Steven Pergam got the Pfizer vaccine: Building trust in safe and effective COVID-19 vaccines (fredhutch.org)
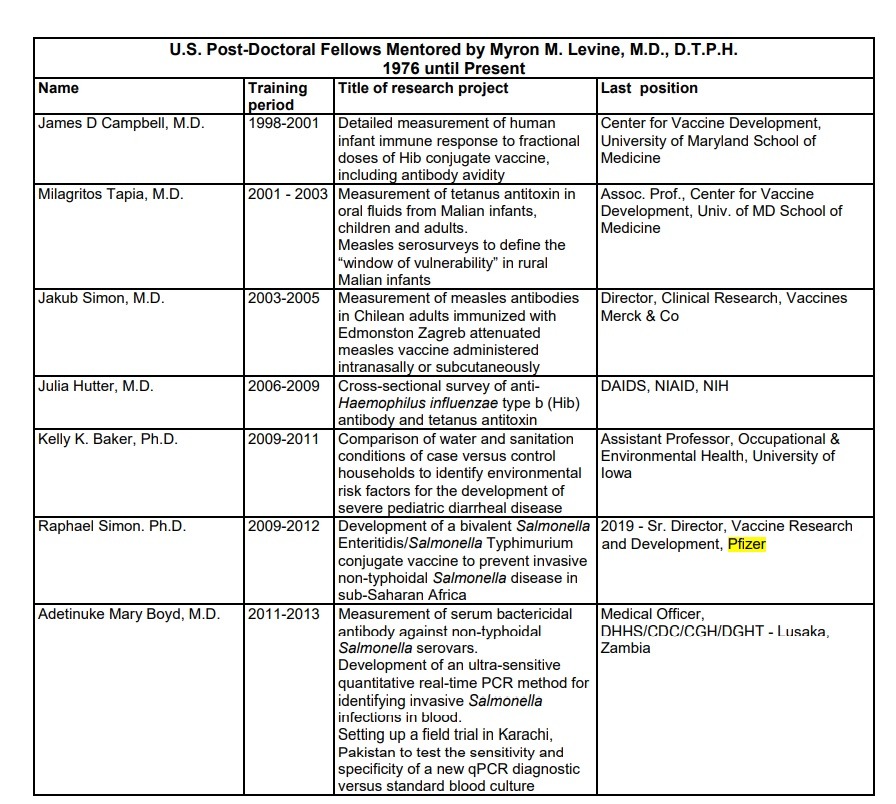
Committee member Archana Chatterjee worked on a research project related to vaccines for infants between 2018-2020, and the research project was sponsored by Pfizer.

Myron Levine has mentored some U.S. post-doctoral fellows, and one of his proteges happens to be Raphael Simon, the senior director of vaccine research and development at Pfizer.
James Hildreth, temporary voting member, made a financial interest disclosure for this meeting in which he disclosed more than $1.5 million in relevant financial interests, including his work as president of Meharry Medical College, which administers Pfizer Coronavirus vaccines.
Geeta K. Swamy is listed as the chair of the “Independent Data Monitoring Committee for the Pfizer Group B Streptococcus Vaccine Program,” a committee sponsored by Pfizer. Duke University states that “Dr. Swamy serves as a co-investigator for the Pfizer COVID-19 vaccine trial.”

Gregg Sylvester previously served as a vice president for Pfizer Vaccines, where he launched Pfizer vaccines including one for children.
 Among the meeting’s “temporary voting members,” Ofer Levy, Boston Children’s Hospital, is for the Pfizer vaccine for children, Eric Rubin is pro-vaccine for children, Jay Portnoy supports authorizing Coronavirus vaccines for kids, and Melinda Wharton complained over the summer about how orders for the CDC’s “Vaccines For Children” program dropped.
Among the meeting’s “temporary voting members,” Ofer Levy, Boston Children’s Hospital, is for the Pfizer vaccine for children, Eric Rubin is pro-vaccine for children, Jay Portnoy supports authorizing Coronavirus vaccines for kids, and Melinda Wharton complained over the summer about how orders for the CDC’s “Vaccines For Children” program dropped.
FDANews stated last December: “FDA advisory committee members in the past have frequently been the target of heavy politicking by industry representatives of whatever drug they were considering for a recommendation at in-person meetings. That process has been somewhat altered by the fact that during COVID-19, meetings are being held virtually. But it’s likely that behind-the-scenes pressuring still goes on. The industry defends the attempts to influence committee members as simply efforts to best present their case.”
Health
FDA Panel Backs Pfizer Shot For Kids: “We’re Never Going to Learn About How Safe This Vaccine Is Unless We Start Giving It”
The same FDA panel approved the rollout of boosters earlier this month based off “gut feeling” rather than data.
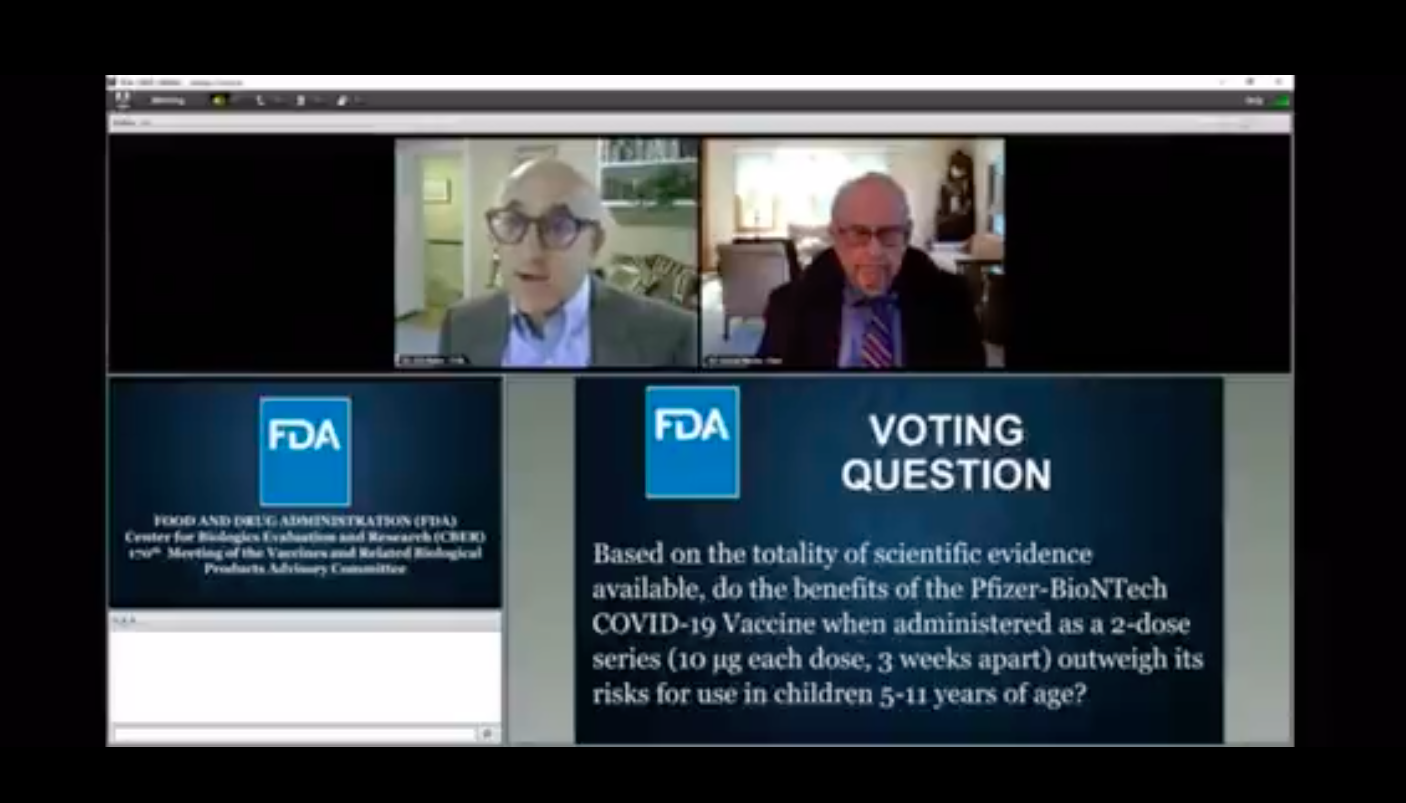
An FDA vaccine advisory panel on Tuesday voted unanimously 17-0 in favor shooting up kids aged 5-11 with Pfizer’s experimental mRNA injection with panelist Dr Eric Rubin stating, “we’re never going to learn about how safe this vaccine is unless we start giving it.”
Full context:
“We’re never going to learn about how safe this vaccine is unless we start giving it,” Dr Rubin said, urging other panelists to vote for it. “That’s just the way it goes.”
The panel voted in favor of experimenting on tens of millions of helpless children with zero long-term data on side effects because 94 children between 5 and 11 have died with COVID-19 (they claimed “of”) and “all have names. All of them had mothers,” to quote the emotional gobbledegook uttered by panelist Patrick S. Moore.
From The Washington Post:
“To me, it seems that it is a hard decision but a clear one,” said Patrick S. Moore, a University of Pittsburgh microbiologist and committee member. He noted that 94 children between 5 and 11 have died of covid-19, and “all have names. All of them had mothers.”
As the WSJ reported:
Members of the FDA’s vaccine-advisory panel supported Moderna’s booster dose even though the evidence for it was from a small study and had mixed results.
“It’s more a gut feeling rather than based on really truly serious data,” said Patrick Moore, a member of the committee and a professor of molecular genetics and biochemistry at the University of Pittsburgh School of Medicine. “The data itself is not strong, but it is certainly going in the direction that is supportive of this vote.”
This is how they “follow the science.”
Health
Governor takes over state’s PRIVATE businesses, mandates vaccines for all
‘His message was crystal clear, obey or lose your job’
The governor of Washington has begun a process that could result in a statewide mandate for all workers to accept the experimental COVID-19 shots in order to be able to get a paycheck.
Across America already, universities, schools and hospitals have COVID vaccination mandates – even though as experimental treatments those actions remain under court challenge in many cases.
President Biden also has ordered the vaccinations for federal workers and the military. And companies with more than 100 employees.
But now Democratic Gov. Jay Inslee in Washington has moved the agenda even further.
A report at the PostMillennial explains under Inslee, the Washington Department of Labor and Industries has proposed an emergency package of rules addressing the “emergency powers” given Inslee to respond to COVID-19.
“This package, which is an extension of current mandates, grants Inslee the ability to enforce COVID vaccine mandates on all private businesses in the state of Washington, according to elected officials,” the report said.
It was a statement from Republicans Jim Walsh and Jesse Young of the legislature that revealed the actions.
“This mandate from L&I demonstrates a complete lack of transparency, which dilutes public trust in our government and fails to show the agency’s good faith in promulgating the rule. The reality is this move by L&I is a blank check for the agency to enforce any of the governor’s mandates or edicts on private employers,” they wrote.
They explained if the state agency wants such a rule, officials should request it in “an open and transparent manner that allows public review and comment.”
“Even if an opportunity for review and comment is not afforded the public, L&I’s website should host the proposed rulemaking to grant easy access to the public,” they said.
They also warned about arbitrary enforcement because of the proposal’s vagueness.
“There is no clear case for ‘good cause’ or ‘the preservation of the public health, safety, or general welfare’ as the governor’s proclamation already addresses these issues, making L&I’s mandate arbitrary and capricious,” they said. “We call on the governor to immediately repeal this mandate. If L&I wants to push this policy, it needs to go through the proper channels and work with the Legislature.”
The report explained Inslee’s recent vaccine mandate was unlike others in that it provided no opt-out for testing instead.
“His message was crystal clear, obey or lose your job,” the report said. “Despite mass protests across the state with thousands of state workers in attendance, Inslee followed through on his orders and terminated thousands that decided not to comply.”
The report warned, “If Inslee follows through on L&I’s emergency rulemaking package, all businesses and employees in the state of Washington will be subjected to submitting proof of vaccination as a condition of employment.”
Real Clear Policy earlier explained that Americans simply don’t like Biden’s orders and mandates.
The article pointed out that health authorities have openly misled the public, including top medical adviser Anthony Fauci’s multiple flip-flops on COVID issues.
He deceived the public, for example, by saying the public didn’t need to wear masks, then again when commenting about “herd immunity.”











